Blood ties: The inspiration behind a potential sepsis breakthrough
Robert Hancock and Olga Peña—who lost her father to sepsis—may have found a way to diagnose the deadliest syndrome you’ve never heard of
Sepsis. Composite coloured scanning electron micrograph (SEM). Bacteria amongst the red blood cells from the circulatory system. When a strain of harmful bacteria gets into the bloodstream, the resulting infection is called bacterial sepsis, or bacteraemia (commonly known as blood poisoning). Multiplication of the harmful bacteria in the blood stream can cause death. When there is a high amount of toxins produced by bacteria in the blood, a septic shock may occur. Septic shock is a highly dangerous condition in which body tissues are damaged. This kind of infection can be serious, but it can usually be successfully treated with antibiotics, if caught early.
Magnification: X5000 when printed at 10cm wide. (Steve Gschmeissner/ Science Photo Library)
Share
The doctor suspected 70-year-old Augustin Peña had the flu, so he sent him home to rest and drink lots of fluids. But after three days of continuing to suffer, Peña entered the local hospital in Tolima, Colombia. His entire body became inflamed and swollen, his skin stretched so tightly that his blood was visible beneath his skin, like magma bubbling at a crack in the Earth’s surface. He suffered severe diarrhea and multiple heart attacks until, one by one, his organs began to shut down. Eleven days after being mistakenly diagnosed with the flu, Augustin Peña died of a little-known illness called sepsis.
Augustin’s youngest daughter stood by his bedside throughout his ordeal. A graduate of Pontificia Universidad Javeriana in Bogotá with a degree in bacteriology, then-23-year-old Olga Peña—like most people—had never heard of sepsis. She could never have imagined that just a few years after her father’s 2003 passing, she’d make a scientific breakthrough that could revolutionize the diagnosis of one of the world’s deadliest syndromes.
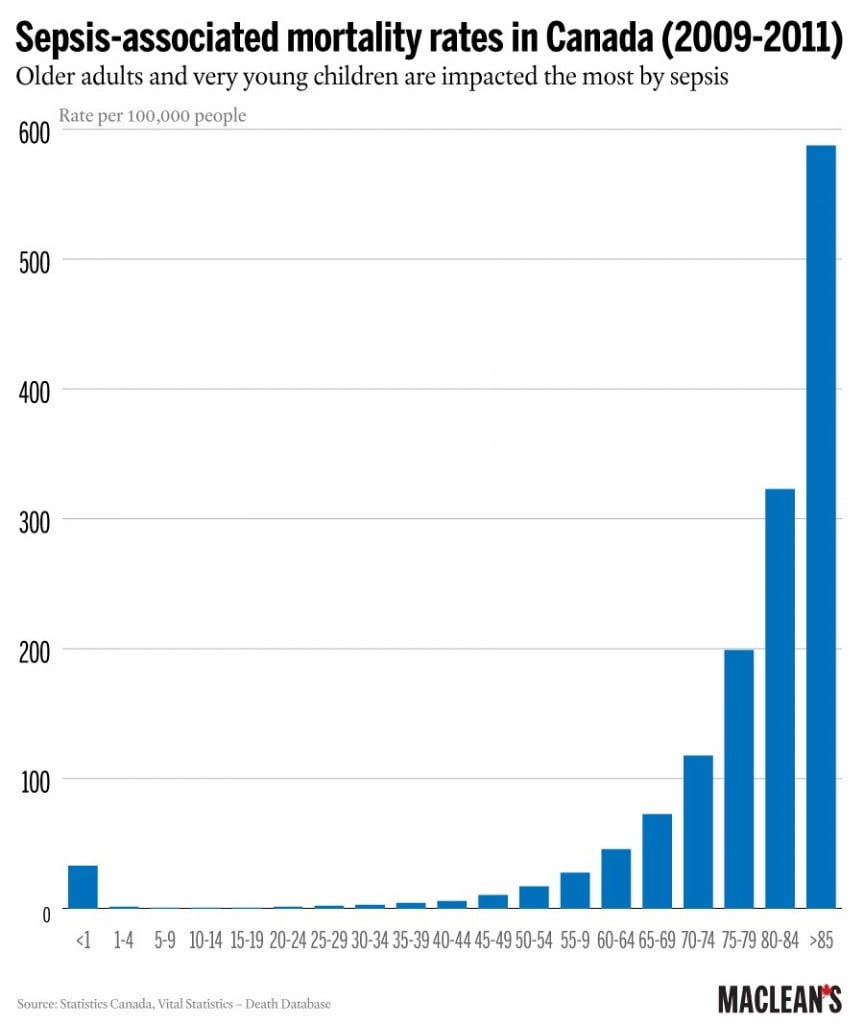
Sepsis—colloquially known as blood poisoning—is not all that rare. In Canada, close to 10,000 people die each year from it, and it accounts for almost 11 per cent of hospital deaths while being implicated in one of 18 Canadian deaths overall. The syndrome is starting to gain more recognition worldwide, with the World Health Organization establishing sepsis as a global health priority in May.
Simply put, sepsis begins with an infection anywhere in the body—from the lungs to the skin to the urinary tract—that gets out of control. In an attempt to fight it off, cells release proteins and chemicals, causing inflammation. This is normal. If the infection goes unrecognized or untreated, however, the entire body can enter an inflammatory state. Eventually, organs stop receiving adequate amounts of oxygen and one by one they begin to shut down. This contributes to about 55 per cent of sepsis cases in Canada. The remaining 45 per cent strike people with a weakened immune system, most frequently because of cancer or diabetes.
The treatment for sepsis is simple: antibiotics and fluids. But think of a building on fire. At first, the fire is in only part of the building, and if firefighters arrive quickly enough, they can contain it. But once the flames have spread throughout the building, it’s too late—the building will burn until it collapses into rubble. With sepsis, every hour without antibiotics increases the risk of dying by as much as eight per cent. This means that if you develop sepsis at noon but aren’t given antibiotics until 6:30 p.m., you have a 50-per-cent chance of dying. With sepsis, a timely, accurate diagnosis is as good as a cure. Making the diagnosis, however, is not simple at all.
“There is no blood test, no imaging test that’s going to 100-per-cent diagnose someone with sepsis,” says Steve Gauthier, a general internal medicine resident at the University of Toronto. “It’s a combination of the clinical picture that the patient presents and their blood work and their history, their physical exam—there might be 10 or 15 factors to consider.”
Step 1: Initial Infection
Sepsis stems from an infection–any infection, although usually bacterial. This could be infection of the skin, the lungs, the bladder, etc.
Step 2: Immune response
In response to the infection, cells release chemicals that cause inflammation. This is normal.
Step 3: Irregular immune response leading to sepsis
The immune system becomes dysregulated and goes into overdrive. It causes inflammation throughout the entire body, and eventually your body fails to recognize the bacteria as foreign. This is sepsis.
Fever, weakness, rapid heart rate and breathing along with an increased number of blood cells signal the onset of sepsis
Step 4: Septic shock
Septic shock is a subset of sepsis and is life threatening. Multiple organs stop functioning, and blood pressure drops dangerously low.
Shortly after her father’s death, Peña moved to the United States where, despite her desire to work in immunology, she ended up as a cancer and genetics researcher. Her father—an elementary school graduate—had always stressed the importance of school, so when Peña’s husband floated the idea of moving to Canada, she agreed, provided she could find a PhD position in immunology.
She contacted more than 100 professors across the country asking about potential projects, but then a researcher named Robert Hancock in Vancouver, offered her a project she couldn’t pass up: the chance to solve the riddle of sepsis.
“It’s not like from the moment my dad died I was thinking, ‘Okay, I am going to find a cure for sepsis,’” says Peña. “This project just felt like an opportunity coming out of the skies, and it was my chance to give back in some way to the knowledge in a field that desperately needs to advance.”

A professor at the University of British Columbia, where he is director of the Centre for Microbial Diseases and Immunity Research, Hancock is considered a world leader in his field. In an interview, he points out that he’s fortunate to get a lot of funding, which gives him the freedom to work on whatever he finds interesting. However, there was one project he couldn’t get any money for, so he paid for it out of his own pocket. The goal was to determine exactly what happens at the cellular level when someone develops sepsis—something no other researcher had been able to accomplish, and something he had been curious about for years. He offered the project to Peña.
Before officially accepting her as a graduate student, Hancock paid for Peña to fly from Los Angeles, where she was working, to Vancouver. Her first trip to Canada was far from easy—and in a way, it would foreshadow the next five years she would spend working on sepsis.
It was November 2006, and Vancouver—a city not well-accustomed to snowfall—was in for a massive storm. UBC shut down completely, and while Peña managed to get to the campus, she got lost once she was there. She eventually found one of Hancock’s students outside and followed him to the office, surprising Hancock, who had been out in the snow searching for her.
“She had worked out how to go from a building over there with no power and snow all over the damn place right into my office,” says Hancock. “And I thought, ‘That’s the sort of person you need in science.’”
Having secured her position in the lab, Peña started work on a topic she had no previous research experience with. She was about to learn the magnitude of the syndrome responsible for her father’s death.
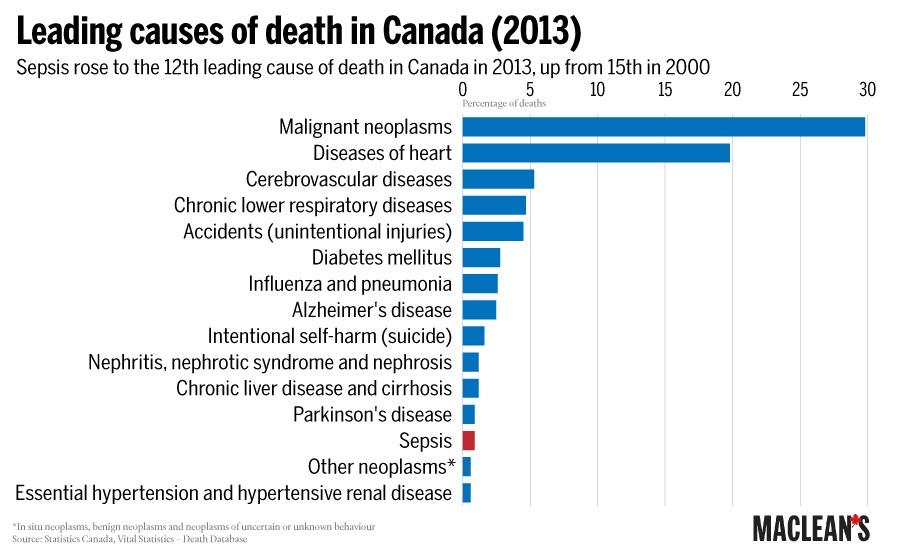
In the United States, there are an estimated 258,000 sepsis-related deaths each year, making it the third biggest killer behind cancer and heart disease. The World Health organization considers sepsis one of the biggest killers of newborns, causing one-quarter of baby deaths worldwide each year.
But all the statistics, including the Canadian ones, are just rough estimates; in fact, they are likely underestimates. That’s because data is often collected from death certificates that don’t have a box to check for sepsis. Instead, physicians are given a few lines to write out the cause of death. Because sepsis always develops as a result of an infection, anyone who dies of it has another medical condition. Most of the time, that other condition is listed on the death certificate. Even when the legendary boxer Muhammed Ali died from sepsis in June 2016, most news reports attributed his death to a longstanding illness, with no specific mention of sepsis.
“Some people might just put pneumonia [on the death certificate],” says Gauthier. “Some people might put lung cancer. Some people might put sepsis caused by pneumonia or caused by lung cancer. It’s not extremely clear and prescribed in terms of the way that you fill these things out.” Compounding the problem is the fact that sepsis itself is often incorrectly referred to as a blood infection or blood poisoning.
Because of its obscurity, sepsis also lacks a strong lobby group, Hancock says. “Things like hepatitis, SARS, swine flu—people pay attention because they have this kind of fear factor. But if you look at all the things people have been worried about for forever, the total number of people who died from these things is the same number of people who have died in just three days from sepsis.”
When Hancock started the project in 2007, 31 different trials had been done by other researchers in failed attempts to understand and treat sepsis. This was the challenge Hancock entrusted to Peña.
“Sepsis is considered to be the graveyard of biotechnology and now is essentially avoided because it is considered to be such a complicated disease,” says Hancock. “It’s like trying to make a therapy for stage-four cancer. You know that people are going to die and your chances of reversing that are low, so why even try?”
But Peña spent a year and a half trying to understand what a person’s cells were doing when they contracted sepsis. And she came up with nothing—not a single one of her experiments yielded a concrete result.
Feeling panicked as her PhD comprehensive exams approached in 2009, she started reading even the most obscure reports she could find on sepsis. She would sit at her kitchen table for hours writing and drawing diagrams until slowly, she started to develop a new idea—an idea so profound it jolted her awake one night as she thought, “Oh my God, this is what’s actually happening!”
Peña hypothesized that during the early stages of sepsis, cells undergo a process known as endotoxin tolerance. An endotoxin is the part of bacteria that triggers the immune system to respond. But if exposure to an endotoxin persists, the immune system will start failing to recognize the bacteria as a threat, a phenomenon scientists have dubbed “immune amnesia.”
To test this theory, Peña identified an endotoxin tolerance gene signature. A gene signature is like a fingerprint: it allows you to specifically identify something that is going on in the body—in this case, if and when endotoxin tolerance was happening.
Her next step was to recruit 72 patients from a hospital who were suspected to have sepsis but who had not been officially diagnosed with it. Peña and her team then took blood samples and tested for the gene signature. Of the 72 patients, 37 went on to be diagnosed with sepsis. In all 37 patients, the signature was present, confirming that their cells were undergoing endotoxin tolerance. The remaining 35 patients who did not end up having sepsis did not express the endotoxin gene signature.
The implication of Peña’s hypothesis was far more significant than she initially realized. It was now 2012, and the resulting gene signature didn’t just provide Peña with a better understanding of sepsis; it also offered itself as a potential diagnostic test. By testing for the gene signature, medics could identify early on whether a patient would go on to develop sepsis—and, most crucially, could do so quickly.
The potential impact of a sepsis diagnostic test is huge, mainly because the biggest obstacle with this syndrome is actually making the diagnosis. “Early sepsis is easy to treat but difficult to recognize or diagnose, and then late sepsis is easy to diagnose, easy to see, but it’s difficult to treat,” says Gauthier.
On Sept.13—also known as World Sepsis Day—in 2016, Peña and Hancock, along with the Centre for Drug Research and Development, a Vancouver-based company that transforms basic health research into commercialized products, launched Sepset, a company focused on developing the sepsis diagnostic tool and implementing it as standard medical practice.
“I think this discovery was made before its time, and I think it’ll take a while to set in,” says Hancock. “But I believe in it. I believe in the discovery. I don’t believe it’s chance. I trust the people who did the work.”
That day, in an unrelated development, the B.C. Sepsis Network launched a sepsis toolkit to provide hospitals with more information on quickly diagnosing and treating sepsis. Despite its authoritative name, the toolkit is about as basic as anything in a hospital can get—a piece of paper containing a simple flow chart with tick boxes. The toolkit reminds physicians and nurses of the possibility the patient has sepsis. The flow chart lists signs and symptoms, and the laboratory tests to order, and reminds clinicians to continue monitoring the sick person.
Dr. David Sweet, clinical lead with the B.C. Sepsis Network, estimates that to date, 56 emergency rooms across British Columbia are working to implement the toolkit. Other similar concepts exist, including an evaluation for severe sepsis screening tool created by the Surviving Sepsis Campaign. The Ottawa Hospital Research Institute also recently put into action a new approach to sepsis care, including an education campaign and the use of a toolkit similar to Sweet’s.

It is possible that the confusion and lack of progress on sepsis is about to be abolished, or at least significantly diminished. But some have their doubts. Even with measures like implementing a sepsis toolkit, Sweet says a diagnosis still boils down to clinical judgement.
“Almost everyone in the emergency room has abnormal vitals,” he says. “So if two of the criteria [in the toolkit] are high respiratory rate and high heart rate, you get those even if you’re nervous or have done some exercise. So if you’re so hypersensitive you’re getting a page of blood work for everybody that shows up in the emergency room, that’s just a waste of time and money.”
As for Peña and Hancock’s diagnostic test, Sweet says this: “If you showed me a data set of 10,000 patients, and you said 100 per cent of the time this test predicted whether someone would develop severe sepsis, I’d be like, ‘Sign me up, that’s the dream,’ ” says Sweet. “The problem is very few things are that good.”
Gauthier echoes this hesitation, pointing to the diagnostic test’s potential limitations. Sepsis patients are often treated with broad-spectrum antibiotics. However, certain infections require treatment with specific antibiotics, something the test won’t be able to determine. And then there is the increasing problem of antibiotic resistance, which could be an issue in treating sepsis in the long term.
“People hate to have new things, like they hate the novelty of this idea,” counters Hancock. “I don’t think physicians really understand that this is a test to tell them someone has sepsis at a time where they have no knowledge at all, just clinical suspicion. It’s going to be important.”
Despite his confidence, Hancock acknowledges that the current test might need some refinement. The next step is to do a bigger trial—one trial of 72 patients won’t cut it when it comes to introducing a diagnostic test that must be consistently accurate in a wide range of populations and medical settings. Hancock is in the process of setting up trials around the world involving thousands of patients, physicians and researchers. He and Peña hope to start their worldwide trial in August. Hancock is actively searching for a partner to help him develop and distribute the test when the time comes—which probably won’t be for at least another four years. Ideally, the finalized test will make a sepsis diagnosis in under an hour; existing alternatives can take up to a few days.
“You have very few opportunities as a researcher to change the world,” Hancock continues. “So if this test works out I would change the world—that’s pretty friggin’ amazing.”
Meanwhile, for now-37-year-old Peña—who was recently promoted to manage continuation of the project—the results of their work are even more personal, and she is hopeful that they will make a difference.
“It’s not my project anymore. It’s out there to serve people that need it so that they don’t have to go through what my dad went through,” she says. “If my dad had been diagnosed within the first two days of being in the hospital, he probably would have had a much better chance of survival.”