Inside Frank Plummer’s brain
The legendary scientist turned to highly experimental deep brain stimulation to treat his own alcoholism. He’d devoted his whole life to science, he joked. Why not his brain, too? The research he took part in is revolutionizing how we treat the world’s most stigmatized illnesses.

Plummer was a leading expert in infectious diseases; his death is a significant loss for Canada as we navigate this pandemic, says Kennelly (John Woods/CP)
Share
The first time Jo Kennelly met Frank Plummer, she barely noticed him.
It was 2006. She was the Ottawa-based policy director to Tony Clement, then the federal minister of health, and she and Clement were in Toronto attending the International AIDS Conference as Canada’s representatives. Plummer, scientific director of Winnipeg’s National Microbiology Lab and one of the world’s top experts in infectious diseases, was the scientist assigned to the federal team. But Kennelly, a New Zealander who did her Ph.D. at the University of Cambridge on HIV/AIDS, was distracted by all the other big names at the event, which drew 24,000 people from around the world.
“I was fangirling everyone else at the conference and had no idea who this tall, lanky scientist was,” she tells me over Zoom in June 2020, smiling crookedly into the camera from her living room in Toronto.
She’s enjoying the joke, which is that she eventually got to know Plummer very well indeed. They fell madly in love and married seven years ago. She calls him the love of her life.
“We were just good together,” she tells me. Plummer, who was wryly funny, understood what Kennelly calls her special brand of “crazy.” Being married to him was “like docking a ship in a safe harbour,” she says.

As we speak, she frequently, fiercely dashes away tears with her fingers. Plummer died in February. She is still surrounded by him. Pale-skinned with fine white hair cut in a side-parted bob, Kennelly proudly takes me on a virtual tour of what she calls “Frank’s corner.” Walls of framed honours and a handsome black bookshelf filled with prizes and plaques for his work on HIV/AIDS and other diseases. An Order of Canada. Four honourary degrees. A Canada Gairdner Wightman and a Killam, which are two of the top prizes available to Canadian scholars. Fond letters from heads of state for his work saving the world from deadly infections. These are just a sampling of the 10 boxes of awards she has packed away.
Kennelly jokes that she and Plummer chronicled their love story by whichever virulent global infectious disease he was trying to keep under control at the time. He declared his love just as the deadly H1N1 flu pandemic struck in the spring of 2009—in between urgent calls with the World Health Organization, the Mexican government and the Canadian Prime Minister’s Office. By the time H1N1’s second wave was cresting that fall, she realized she loved him as well—over a Thanksgiving dinner he contrived to cook for her and her three children in his hotel suite in Ottawa.
But the legendary scientist was also secretly addicted to alcohol. And that, combined with a genetic predisposition to liver disease, put his life in danger. He spent several years going through every treatment on offer, including a new liver. Nothing worked.
Then one day, in 2018, his addictions specialist mentioned a highly experimental clinical trial at Sunnybrook Health Sciences Centre in Toronto. Deep brain stimulation involves permanently inserting a pair of electrodes into the brain and then running an electrical current through them from a battery pack nestled near the collarbone. The hope was that the current would interfere with the part of the brain making Plummer crave alcohol, but it was so early in the investigative process that the questions were legion. Was it safe? Would it work? How else would it affect his brain?
Plummer plunged into the scientific literature, reading everything he could lay his hands on. It was new. It was unknown. For his whole career, he’d been the one in the lab coat taking notes. Why not reverse the role? “He joked that he’d committed his whole life to science and so he was literally committing his brain this time,” Kennelly says.
He decided he wasn’t worried about side effects or complications, and neither was Kennelly. “He was worried about a horrendous death from alcohol,” she says. In December 2018, Plummer lay down on an operating table at Sunnybrook and became the first person in North America—and possibly the world—to have electrodes inserted into his brain to control his alcohol use disorder (the term used by Sunnybrook) as part of a clinical trial.
And then he waited to see what would happen.
***
Welcome to the brave new world of the brain. Scientists are reimagining some of the most stigmatized mental illnesses, such as drug addiction, depression, obsessive-compulsive disorder (OCD) and post-traumatic stress disorder (PTSD), as wiring problems. It’s a fundamental reframing from the old days of pegging these illnesses to, say, chemical imbalances that need to be regulated with medications, or personal choices that require sterner self-control, or even psychodynamic disorders rooted in family-of-origin issues that only years of analysis can mend.
The idea is that electric activity in specific parts of the brain affects a person’s thoughts and feelings and actions. Sometimes bits of the circuitry become supercharged, whether by trauma or substance abuse or genetics or something else, making a person feel and do things that can hurt them. And sometimes, medication and talk therapy can help rewire those circuits. But when nothing else works, performing brain surgery to jam or sever the tiny electrical circuits responsible for those thoughts might make the person feel better. Or so the thinking goes.
It’s a new scientific discipline called neuromodulation. Three of the most keenly studied techniques are transcranial magnetic stimulation, focused ultrasound and deep brain stimulation. Transcranial magnetic stimulation involves repeatedly putting magnets on the outside of the head to temporarily interrupt brain circuits with waves from a magnetic field. Focused ultrasound permanently severs a tiny piece of the brain’s electric circuit with a targeted beam of high-frequency sound waves. Deep brain stimulation, which is what Plummer had, involves surgically inserting a pair of thin electrodes into the brain that give off a continuous or intermittent current of electricity, essentially shutting down that part of the brain.
READ: The story behind a vegetative patient’s shocking recovery
The techniques are not brand new. Deep brain stimulation, for example, has been widely used for a couple of decades to treat tremors. But using this suite of techniques on psychiatric illnesses is novel—and still highly experimental. Clinical trials are in the works at dozens of hospitals internationally, and Canada is leading the way. Sunnybrook alone has more clinical trials than anywhere else in the world, and Toronto Western Hospital has also long been in the vanguard.
“I always considered psychiatric disorders as no different from any other brain conditions—strokes or brain cancer,” says Nir Lipsman, the neurosurgeon and neuroscientist who is director of the Harquail Centre for Neuromodulation at Sunnybrook. “The only difference is that with those conditions you can see something wrong on a [brain] scan. With OCD or a major depression or schizophrenia, you often can’t.” Lipsman did Plummer’s brain surgery and is the driving force behind Sunnybrook’s multiple trials of new neuromodulation treatments.
But the experimental procedures under investigation have come with an intense focus on how they could—and should—be used in the future. “The questions are: what don’t we know and why don’t we know it?” says Jenell Johnson, a professor of communication arts at the University of Wisconsin-Madison who has written extensively on the history of psychosurgery. “There’s so much in psychiatry people don’t know. People call the brain a black box for a good reason.”
The practice of physically modifying the brain, and therefore controlling emotions and behaviour, has had a troubled track record over generations. Electroshock treatments. Ice-pick lobotomies. Permanent implants in the brain. These have long been a staple of science fiction scripts in which villains take control over the minds of unwilling victims. Fiddling with the nuts and bolts of the brain has come to symbolize the epitome of authoritarian control.
“The brain is the seat of our identity. It’s the seat of our memories. It’s who we are. It’s what makes us us. Any intervention that threatens that, or potentially does, or puts that at risk, is going to be greeted with a visceral response: ‘Well, is this the right thing to do?’” says Lipsman. “And I think appropriately so.”
It raises the question: how do we harness these new techniques to heal, but make sure they do not harm?
***
Alcohol addiction is a remorseless foe. It got its first grip on Plummer when he was based in Nairobi 25 years ago and never quite let him go.
It began with tragedy. He had landed in Kenya at the beginning of the global HIV/AIDS epidemic and was treating infected sex workers in Nairobi’s Majengo slum when he discovered some who had natural immunity. That held out clues about how the virus is transmitted and offered hope that one day there could be a vaccine. By 1995, his work was famous, and he and 25 colleagues went to present it at an international conference on AIDS in Kampala, Uganda. Five of his Kenyan colleagues were killed in a car crash on their way home.
Plummer was devastated. Back home in Nairobi, he began drinking Scotch after work. And crying. He drank on the long flights back to Canada. “Scotch was my friend and my way of communicating with myself,” he wrote in a Sunnybrook blog after his surgery. “It would put me into a pleasant trance, giving me the mental space to reflect, dream big, and it made the journey seem shorter.”
The alcohol didn’t impair his cognitive skills. “With addictions, as I’ve understood it, people can be very, very high-functioning,” Kennelly says. “Frank got up every morning and went to work and ran on adrenalin all day.”
Eventually, Plummer, who stood six-foot-four, drank as much as 20 ounces of Scotch a night, never showing signs of intoxication. He didn’t realize he had a problem until 2012, when his liver gave out. His mother had died at 52 from non-alcoholic liver disease, which he had inherited. For two years he hovered between life and death, also battling cancer of the colon and kidney, until he had a liver transplant in 2014. Plummer and Kennelly sailed through all of it: addiction, rehab and hospitalization. “We just scheduled it into our day,” she tells me.
He bounced back from the transplant and had a long period of sobriety. But eventually, he couldn’t control his yearning for alcohol. That’s when it became clear to him that it was a full-fledged addiction rather than just a source of comfort. “Every time he would slip was so tough for him. Because when you slip, there’s the shame,” Kennelly says.
His second liver began to falter. He went into rehab and did counselling. The craving continued. He was dying.
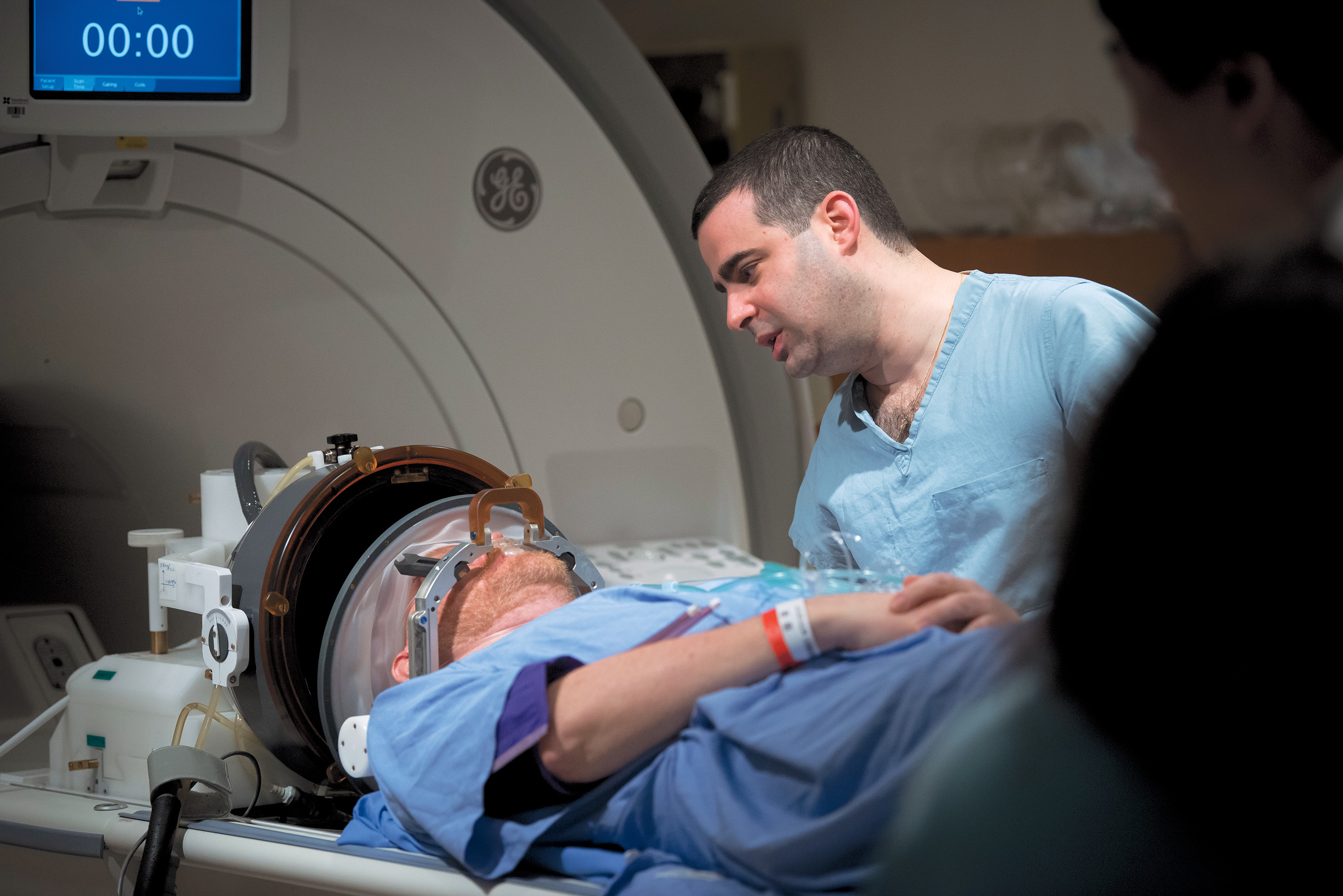
Then came the decision to have brain surgery. On the operating table, Lipsman attached a frame to Plummer’s head and conducted a brain scan to assign three-dimensional coordinates—x, y and z—to every spot of Plummer’s head. He drilled two small holes in his skull while Plummer was under anaesthetic, then woke Plummer up as he placed a wire electrode just 1.5 mm thick into each side of the nucleus accumbens, the part of the brain involved in processing reward.
At that point, he turned the electrodes on.
“It never gets old,” Lipsman tells me. “You’re at the interface of mind and body right at that moment.”
Plummer felt different as soon as the current began to flow, right there on the operating table. He was happy, even chatty—a shift from his usual restrained manner.
Because it was such an early-stage trial, one of its principal goals was to see whether the procedure could be done without complications such as hemorrhage or infection. Plummer was all clear on that front. But Plummer and Kennelly eagerly tracked other changes, too, recording their observations for posterity. His mood improved. He began to get up early. He was ferociously productive.
“The craving at the back of your brain that you’re constantly trying to satiate but you can’t satiate had gone away,” Kennelly says. He was not fully cured, but there was a transformation in him. For the first time in a long time, Plummer was hopeful.
***
Today’s neuromodulation techniques are descendants of earlier attempts to treat brain illnesses. Thousands of years ago, a common method was trepanning, or cutting holes in the skull, to release pain or pressure or even evil spirits from inside the head. Ancient Egyptians used shocks from electric eels to treat pain and brain disease. By the first decades of the 20th century, doctors were putting patients into repeated insulin comas or using electroshock therapy to treat mental illness.
But the most notorious psychiatric intervention—and the one against which all future procedures will be measured—was the lobotomy. The procedure, which surgically and permanently severs connections between the frontal and prefrontal lobes of the brain, gained acclaim after the Second World War when hospitals were filled with traumatized soldiers. Tens of thousands of people around the world had the operation, many as a last-ditch effort to keep from being incarcerated in asylums.
Doctors and patients originally viewed it as a miracle cure. The Portuguese neurologist Egas Moniz won the Nobel Prize for medicine in 1949 for perfecting it. American neurologist Walter Freeman enthusiastically promoted it with the help of neurosurgeon James Watts. Their technique of anaesthetizing patients, drilling through the skull and disabling parts of the brain became a mainstream treatment for depression, schizophrenia and other mental conditions, and sometimes just for odd behaviour. Freeman once did the operation on a four-year-old.
But while some patients reported relief, results were mixed. There were patients who were severely disabled. Some died. Neurosurgeons only crudely understood the structure of the brain. They could not be precise about where they were cutting. “There was less understanding and more mystery [around] the brain in the past,” says the ethicist Walter Glannon, a philosopher at the University of Calgary.
The depiction of lobotomy and electroshock treatment as a brutal punishment for the character played by Jack Nicholson in the 1975 Oscar-winning movie One Flew Over the Cuckoo’s Nest lives on in the public mind, Glannon says. Even today the abuses of the movie’s evil Nurse Ratched instill fear in people over psychiatric practices, a phenomenon amplified in the recent Netflix series based on her story.
Apart from the fact that it was frequently administered without informed consent, a major detriment of lobotomy was a change in personality. Take Rose Samuel, the pseudonym of one of Freeman’s patients, who was in her twenties when he treated her. Her case is described in the 2013 book The Lobotomy Letters by the medical historian Mical Raz, who had access to Freeman’s archives. Ten days after Samuel’s procedure, Freeman writes: “Rose is a smiling, lazy and satisfactory patient with the personality of an oyster.”
Freeman eventually developed a quicker, cheaper technique that could be performed in an office. Rather than anaesthetizing the patient and drilling through the bone, he would induce a convulsion through electroshock therapy and then insert a spike through the skull’s eye socket into the brain, a procedure now infamously known as the ice-pick lobotomy.
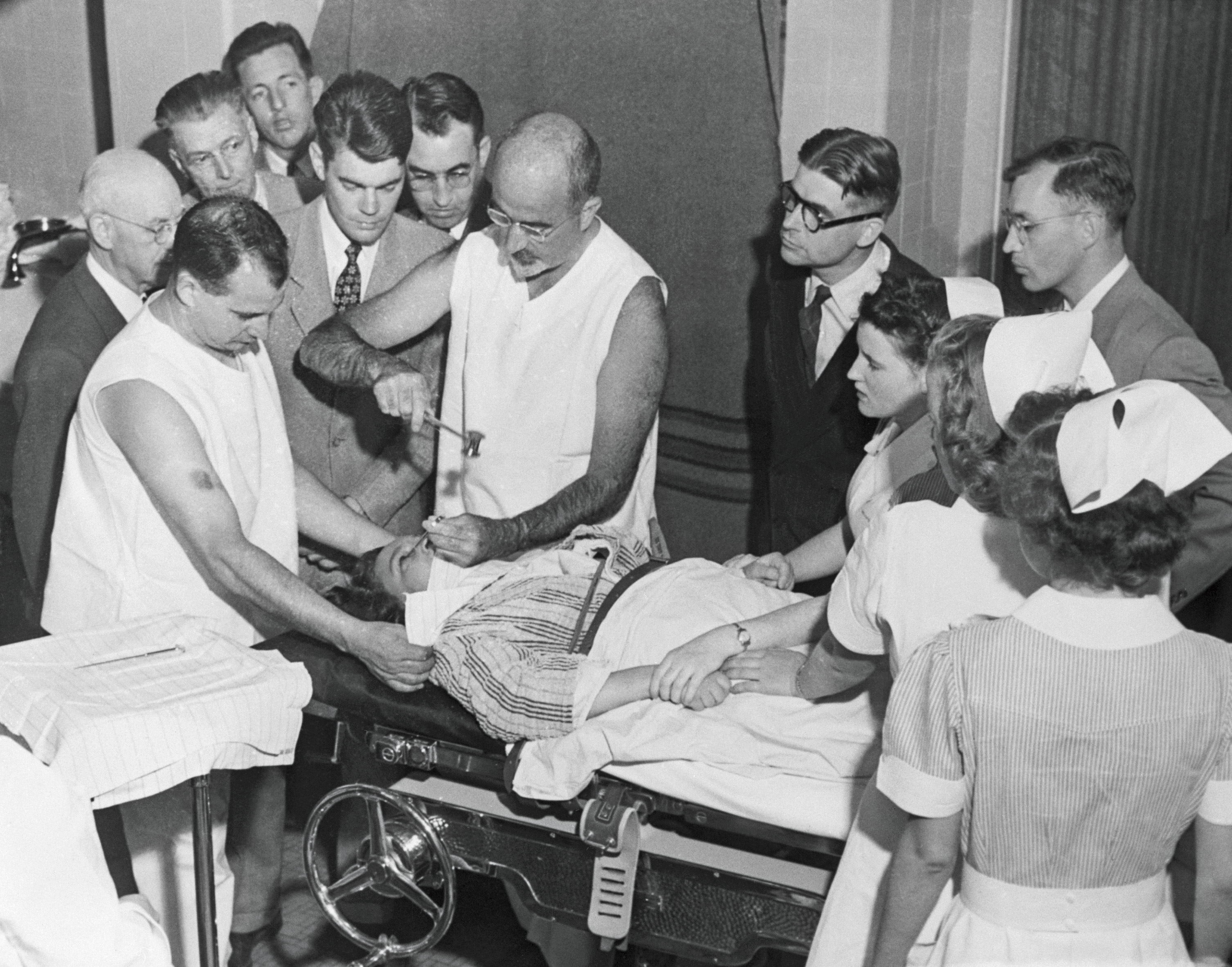
Lobotomies finally fell out of favour with the arrival of the first psychotropic drugs in the mid-1950s. Stimulants and tranquilizers treated depression and anxiety. Antipsychotics came into vogue for delusions, agitation and schizophrenia. By the mid-1960s, the focus had shifted to controlling how much serotonin coursed through the brain. The early drugs were considered a sensational and effective breakthrough to treat mood disorders, explains Edward Shorter, a historian of psychiatry at the University of Toronto, in his book Before Prozac: The Troubled History of Mood Disorders in Psychiatry. Biochemical rationales for mental illness reigned, a belief summed up by a quote Shorter cites from the American neuroscientist Ralph Gerard: “For every twisted thought there is a twisted molecule.”
But alongside the rise of drugs and psychotherapy in the decades that followed, neurologists began taking a closer look at the brain’s electrical circuitry. One breakthrough was in research on electroshock treatments, which deliver electrical energy to the brain and throw it into seizure. At one time, physicians thought that it was the seizure that helped the patient, says Anthony Levitt, a psychiatrist who is chief of the Hurvitz Brain Sciences Program at Sunnybrook and is involved in its neuromodulation program. Then they came to understand that it was the charge of electricity that had the effect of rebooting the brain. That led to the development of magnetic therapies applied from outside the head, which could deliver focused magnetic energy to brain tissue without the need for a general anaesthetic or the side effects of a shock-induced seizure.
At the same time, our understanding of the anatomy of the brain became far more precise through electroencephalography (which records electrical impulses passing among brain cells), computerized tomography scans (a series of X-rays of tissue, bone and blood vessels built into a three-dimensional model), real-time magnetic resonance imaging (MRI) and experiments on animals. These tools allowed neurologists for the first time to map the circuits that relate to such things as movement, memory, emotions and rewards, often down to the millimetre.
And then, about a decade ago, neuroscientists like Kullervo Hynynen, vice-president of research and innovation at Sunnybrook Health Sciences Centre, started to work out how to focus a beam of ultrasound waves through skin, muscle and bone into a targeted part of the deep brain without harming tissue. Guided by MRI, Hynynen and others figured out how to use the waves to heat up a precise part of the deep brain, momentarily stunning it, and then test the results while the patient was on the table. If the results were good, the surgeon could increase the beam’s energy, raising the temperature a little higher to cause a permanent cut in brain tissue. All without the risks of opening the skull. Hynynen is known as the “Wayne Gretzky of focused ultrasound” for his innovations.
Separately, neuroscientists were experimenting with other surgical methods of treating neurological disorders such as epilepsy. They knew that epileptic seizures were caused by eruptions of energy in the brain. They treated epilepsy by surgically severing some of the connections where those discharges of electricity were taking place. But what if the seizures could be controlled by a constant, low-energy pulse of electricity, delivered by electrodes implanted in the brain? A similar quest was afoot to control tremors from Parkinson’s disease. Deep brain stimulation is now a standard way to treat tremors from Parkinson’s and is gaining favour as a treatment for epilepsy.
Much more recently, focused ultrasound began being used to temporarily open the blood-brain barrier, allowing drugs to pass through. Two years ago, Paul Hudspith, 54, the senior vice-president of a global engineering consultancy and a professional cellist who lives north of Toronto, had emergency surgery for a brain tumour that turned out to be glioblastoma, an aggressively lethal form of brain cancer. He received radiation and chemotherapy, and then agreed to be one of the first patients in the world to participate in a clinical trial to deliver chemo to the brain in this new way. His most recent MRI showed no tumour, he tells me in June. “I’m not saying that focused ultrasound is a cure for glioblastoma,” Hudspith says. “What I know is that, for today anyway, I’m well.”
But neuromodulation was overwhelmingly being applied to what are traditionally thought of as physical illnesses. The breakthrough of using it to treat mental illness came in 2005, when the trends of mapping brain circuitry and deep brain stimulation converged in an experiment to treat intractable depression. Neurologist Helen Mayberg, then at the University of Toronto, figured out that inserting thin electrodes into Brodmann area 25 of the brain, a linchpin of the brain’s mood wiring, could disable the area and lift depression. She published a landmark paper on it.
After that, neurosurgeons around the world were galvanized, devising experiments to test the family of neuromodulation techniques on a range of psychiatric and other brain conditions. Some of their early findings were pure serendipity. For example, a patient treated unsuccessfully with deep brain stimulation for obesity reported having vivid memories, which launched a bout of research on stimulating the same part of the brain for Alzheimer’s disease.
Today, deep brain stimulation and transcranial magnetic stimulation are under investigation internationally across multiple areas of the brain for major depression, OCD, PTSD, schizophrenia, anorexia, bulimia, obesity, autism, addiction and pain, among other conditions. Focused ultrasound is in trials for many of the same conditions, as well as for the fatal neurodegenerative disease Amyotrophic Lateral Sclerosis (ALS) and traumatic brain injury. Early results suggest it may also stimulate the growth of new neurons in the brain, a hopeful sign for degenerative diseases such as Alzheimer’s, Hynynen says.
And at Sunnybrook, Lipsman is so eager to explore potential benefits that he developed a strategy to speed up the pace of approvals for new procedures. Instead of researching a single treatment for a single condition, he has launched a raft of serial clinical trials of different neuromodulation techniques across many conditions at the same time. It’s like moving at warp speed.
“That’s a philosophical shift,” Levitt says. “From the outside it may not seem huge, but it’s monumental inside the field of clinical sciences.”
***
Finding new ways to help mentally ill patients who have no other options has become a pressing need. While medicine has clocked dramatic improvements over the past 40 years in treating ailments such as heart disease and cancer, the same can’t be said for psychiatric illnesses, Levitt notes. And yet, those conditions can be just as debilitating. The Centre for Addiction and Mental Health in Toronto calculates that mental illness is a leading cause of disability in Canada, and that its economic burden is $51 billion a year. It’s a similar picture around the world.
About a third to half of people with mental illnesses don’t respond to drug therapy and some who do have awful side effects or stop taking medication and fall back into distress. Then, there’s little doctors can do to help them. “This new notion that the brain is a circuit and the fact that they are circuit disorders gives us new hope,” Levitt says.
It’s also a notion that has led to some resounding successes for the Sunnybrook team. In February 2019, Serena Kelly, 48, became the first patient in Canada to have deep brain stimulation to treat PTSD. She is telling me about it on a Zoom call from her home somewhere in Ontario. She can’t say exactly where because she has long been stalked and harassed by her abusive ex-husband and doesn’t want him to know where she lives. Tortoiseshell glasses and dark brown bangs frame her eyes. She’s asked for the questions in advance and has written out detailed answers. When she speaks, she is precise and deliberate.
She was diagnosed with PTSD nearly two decades ago, after years of sexual, physical and emotional assaults that began when she was 12. Certain smells—the ex-husband’s cologne, for example—and phrases or songs triggered nightmares and flashbacks of the abuse. She was constantly terrified and vigilant. She worked intensively with psychiatrists, psychologists and social workers on both the PTSD and depression, but continued to struggle. The breaking point came when her daughter, Harley, died in a motorcycle crash in 2017. That’s when PTSD took over Kelly’s life. She couldn’t drive or hold down a job. She didn’t leave the house for 19 months. Medications and therapy failed to help.

Then her current husband heard about the clinical trials at Sunnybrook on a television show. Kelly submitted an application and went to see Peter Giacobbe, a psychiatrist and clinical lead at Sunnybrook’s Harquail Centre for Neuromodulation who functions as the gatekeeper for the program. “I’d had years of getting my hopes up and nothing happening,” Kelly says. “I was to the point I felt I had nothing left to lose.”
After her surgery, the PTSD vanished. Her depression remains but has lessened. She’s feeling so good that she’s nearly finished her undergraduate degree and is thinking about a graduate program in psychology. “If you had told me two years ago that I would be back in school, that I would be able to drive on my own, that I would have a life free of PTSD, I would have thought you were crazy,” she says, flashing a grin for the first time.
Jeffrey Kotas, 32, struggled with OCD from the time he was in middle school. Red-haired and wearing a relaxed grey T-shirt and headphones, he talks to me from a room painted white in the house he shares with his parents in west-end Toronto. Behind him is a tidy white bookshelf, with volumes neatly arranged. By the time he reached adulthood, Kotas tells me, the disorder had accelerated, torpedoing his plans for university. Medication helped episodically, but the disorder always came back. Like many sufferers of OCD, he became fixated on contamination and cleaning. On occasion he would clean for 36 hours straight, going through huge quantities of Lysol and paper towels. By the time he got to see Giacobbe, he’d been cooped up at home for a decade.
In July 2017, Kotas became the first patient in North America to have focused ultrasound for OCD, followed by nine weeks of intensive live-in therapy at the hospital. To his joy, the fear and anxiety began to abate. He started to be able to do things that had previously been impossible: use his computer, go shopping, walk in the neighbourhood, make friends, tell jokes and even give his mother a hug. “I felt like I was getting back the life I never had,” he says. “It just completely transformed my life and my whole way of thinking and viewing the world.”
That’s not to say there have been no negative side effects during these clinical trials at Sunnybrook. A few ultrasound patients feel unsteady on their feet for a while or have some numbness or headaches. One deep brain stimulation patient had to have the electrodes removed after infection set in, and another underwent a second surgery so the electrodes could be repositioned. But the side effects have been neither common nor serious, Lipsman says. There’s been no brain bleeding or swelling or neurologic problems in any of the focused ultrasound or deep brain stimulation trials. And, critically, no diminishment in cognitive ability with focused ultrasound, either. That was an open question until recently.
Lipsman is careful to track and report any ill effects. “It’s critical that, as promising as these procedures are, both patients and the public are aware of the potential risks, even if they are relatively rare,” he says.
And fundamental questions remain. For example, while neuroscientists have some clues about why the techniques are working, they’re not precisely sure how. Which cells, which molecules, which systems of the brain are affected? Nor are the techniques a cure. The brain has to learn to function in new ways once a circuit has been cut. That takes therapy, and sometimes drugs, too. And time. “It’s not a light switch, it’s a dimmer,” Lipsman says.
***
All of a sudden, the post-operative Plummer had energy to spare.
He spent mornings writing his memoirs (which included 10 pages on dogs he’d loved). The guy who had once been the quietest person in the room became uninhibited and vivacious. He planned a kitchen renovation. He began making feverish plans to go back to Africa with Kennelly and some of their adult children and to attend a conference celebrating the 40th anniversary of his landmark work there. He even invited his friend Anthony Fauci, the U.S. infectious disease expert, who was forced to decline because of a heavy workload.
And he cooked. “Old Frank” had been a good cook. “New Frank” was a superb one. Every day at noon he went for a walk with the dog to the local butcher shop to find his cut of the day. Then came prep and execution, all conducted “with the level of dedication of a biochemist,” Kennelly says. “If you take away a craving and you have a mind that’s been trained [to crave], you’ve got to replace it with something else.” And while the craving for alcohol was gone, the ingrained habit of drinking wasn’t. Plummer still drank alcohol occasionally, but not nearly as much. And he could stop when he wanted to.
Along with the good came some bad. A diagnosis of throat cancer just weeks after the brain surgery kicked him down for a while. The man who had had a knack for tuning things out now noticed everything. The physician whose entire career had been built on empathy now seemed short of it. He railed at flaws in newspaper articles in impassioned, essay-length letters to the editor. At one of his checkups, Frank told his doctor: “I think I need some help. Jo told me yesterday that she didn’t get me through three cancers, a liver transplant and a brain operation to end up being married to an asshole.”
The amazing result of that revelation is that the medical team simply adjusted the settings on Plummer’s electrodes. The sharp edges of “New Frank” smoothed out. Kennelly jokes that she told her friends their husbands should have electrodes installed, too.
Another profound shift was taking place. From the first conversation with Lipsman and his team at Sunnybrook about the clinical trial, Plummer’s shame about his addiction began to wane. For years, he and Kennelly had heard from rehab counsellors he had a choice about whether to drink. “If society is saying to you, and AA and all the rehab groups are saying to you, ‘Well, it’s his choice whether he stays abstinent or not,’ then you, as a family member, feed into that,” Kennelly says. “You feel like they should be stronger.”
But as he read more about deep brain stimulation, Plummer began to see his craving as a brain circuit malfunction that he couldn’t fix himself. He needed the help of another scientist. “For the first time ever, someone talked about an illness that he had in clinical terms, how the brain reacts to and is changed by a substance,” Kennelly says. “And it all made so much sense to me.”
A year after the operation, the month before the longed-for trip to Africa, Plummer agreed to go public about his addiction and the clinical trial. Kennelly was nervous. “This was the new-brain Frank. He was [saying], ‘Well, what are they going to do? They can’t take my Order of Canada off me. They can’t take my awards off me. They can’t take my publications off me. And I think it’s going to do some good.’ ”
His story became an international sensation. Kennelly was getting calls from relatives in the U.K. about the headlines: “Doc cured from booze,” read a lurid one. She rolls her eyes. “The nuances were lost in the stories.”
***
Today, in order to receive neuromodulation therapy to treat mental illness, you have to be in immense pain. And not just in pain, but also unresponsive to every other treatment over many years. It is the last resort. “These are people who are, in essence, palliative,” says Giacobbe, the Sunnybrook psychiatrist who helps figure out which patients should be in the clinical trials.
Giacobbe’s focus is both to ease their suffering and see whether the techniques work. To him, there’s little room for jumping ahead to future ethical considerations if the science isn’t proven to begin with. “Unless we are able to show that these procedures are helpful and that there’s meaningful improvements, then it’s not going to come to pass that it will be widespread,” he says.
So far, it’s unclear whether the procedures will work reliably enough to become mainstream for untreatable mental illness. Alongside streams of published journal articles on remarkable successes, there have been some flops. A few years ago, for example, two large industry-sponsored trials to treat intractable depression with deep brain stimulation were terminated partway through when researchers found no difference between the group whose electrodes were on compared to those whose were off. The failures threw the neuromodulation community into tortured self-reflection. Was the treatment ineffective? If so, why? Were the researchers looking for the wrong outcome?
More troubling, two small studies in 2006 of patients who had deep brain stimulation for Parkinson’s disease found that even though the tremors got better, three of the 29 patients said they had lost parts of their personality. A 38-year-old woman said her passion for life had left her. “I don’t recognize myself anymore,” she said. One reported: “I’m an electric doll.”
The studies opened a lively discussion among neuroscientists and neuroethicists, perhaps mindful of Freeman’s oyster patient, about whether inserting a device into the brain affects one’s sense of self. And if it does, how does that differ from changes that come from psychotherapy or medication or recovery from mental illness? Should those worries outweigh the possible benefits that come from physically altering the brain?
On the other hand, if neuromodulation proves to be a wildly effective and relatively easy way to treat brain illnesses, some wonder whether patients will press to get it before they are in extreme circumstances. Should they have to wait?
Separate conversations are going on about whether the techniques should be used to treat people who are violent or antisocial. But how does society define what is normal and what is dangerous behaviour? At one time, women who wanted the vote were considered antisocial, and homosexuality was thought of as deviant. Today, people who oppose fascism and white supremacy are being called out as dangerous by some in power, including outgoing U.S. President Donald Trump.
At the same time, neuroethicists are grappling with whether healthy patients should eventually be able to have neuromodulation to enhance their brains. A survey of North American neurosurgeons published in 2011 found that more than half saw no ethical problems with providing deep brain stimulation to people who wanted to sharpen their memories.
When I ask Hynynen, of focused ultrasound fame, what the field will look like in 20 years, he muses about whether we will be able to pump entertainment directly into the brain or erase harmful memories. The scenarios scare him. “Luckily we are not at that point yet,” he says.
Johnson, the professor of communication arts who has written about the history of psychosurgery, says that while there’s plenty of cause to be mindful about the ethics of neuromodulation, it’s also important to think about the fallout of failing to explore possible benefits from these new techniques. “The brain is seen as this very sacred thing,” she says. “Is research being unnecessarily halted because there are ways that it discomfits us?”
The fact that all these complex questions are under impassioned discussion this early in the arc of the research is not an accident, says Judy Illes, Canada Research Chair in Neuroethics at the University of British Columbia. Illes, Lipsman and others recently launched the Pan-Canadian Neurotechnology Ethics Consortium to construct neuromodulation’s ethical architecture, with an eye to how the Human Genome Project of the 1990s transformed medical thought and practice. Fast innovation in psychosurgery could have deep implications, too, if not as far-reaching as mapping human DNA.
And it riffs off huge advances in neurosurgery in the 1970s and ’80s, including new techniques in neuroimaging with computerized tomography scans and MRI. Today, as technical prowess and ethics march together, the possibilities for neuromodulation are immense. “There will be yet another revolution,” Illes says.
***
Plummer should probably have been in the hospital instead of on his way to Kenya. He had a touch of pneumonia, but he was hell-bent on making the trip. He’d even put off work on the kitchen renovation to put more money into the kitty for the trip, Kennelly tells me, chuckling as she pivots the camera to a piece of plywood standing in for the new counter they were planning. It felt like a gift he couldn’t turn down. Without the deep brain stimulation a year earlier, he wouldn’t have been healthy enough to go.
They had a glorious couple of weeks at the conference, touring the Majengo clinic Plummer had worked in, revisiting every house he’d lived in, going on safari. He was even still doing interviews about the deep brain stimulation he’d had. Then, on Feb. 4, while still in Africa, he had a massive heart attack and died.
Kennelly breaks down again. COVID-19 was just emerging while they were away and Plummer had been keeping a close eye on its development. He’d trained generations of infectious disease physicians and launched institutions just for this possibility. There’s a huge toll for Canada in Plummer not being here to help guide us through the pandemic, Kennelly says.

She’s continuing his work by helping to raise money for the Majengo clinic’s new roof and for developing the HIV/AIDS vaccine he was excited about. The University of Manitoba is creating an endowed chair in his name.
Kennelly suspects that he knew he was going to die in Africa, she tells me, wiping away more tears. He said he was in heaven, being back there where his work had made such a difference. It was as if he had come full circle. And, like an elderly man might, he spent some of his time there reminiscing about what he’d achieved, including the deep brain stimulation. “I think an important part of Frank’s humanity as a scientist,” Kennelly says, “was accepting his own fragility and turning to other scientists to help him in that.”
She buried him in Kenya on Valentine’s Day. Elder sex workers of Majengo, now grandmothers, showed up and sang hymns in Swahili as they laid him to rest. Along with a towering scientific legacy in both infectious disease research and deep brain stimulation, Plummer left generations of African women who might have died without him. And a Canadian love story that still catches fire.
This article appears in print in the February 2021 issue of Maclean’s magazine with the headline, “Inside Frank Plummer’s brain.” Subscribe to the monthly print magazine here.